Fibroadipogenic progenitors (FAPs) and skeletal muscle regeneration
Skeletal muscles are capable of regenerating after damage. This is possible owing not only to myogenic stem cells (MSCs), from which new myofibers originate, but also to several non-myogenic cell types resident in muscles. Among these, fibro-adipogenic progenitors (FAPs) are known progenitors of tissue-fibroblasts/myofibroblasts and adipocytes. FAPs, are quiescent in healthy muscles, but are triggered to proliferate in situation of muscle damage, resulting in a transient fibrosis, during which they positively influence the myogenic repair process [1, 2]. Besides secreting pro-myogenic factors, FAP activation/expansion enables the transient production of extracellular matrix (ECM), which provides the adequate tissue-stiffness for MSCs to undergo myogenic differentiation [3, 4].
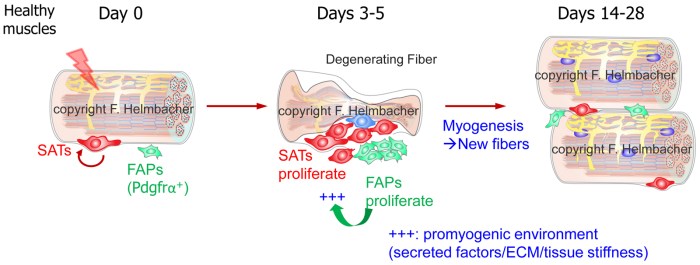
Given their differentiation potential, FAP expansion and differentiation are strictly suppressed in healthy muscles, and strongly limited in situation of skeletal muscle repair, to prevent the appearance of fibrosis and adipose infiltrates, two adverse events occurring in pathological contexts. A first gatekeeper drastically limits their differentiation, while a second mechanism ensures the elimination of activated FAPs present in excess by the innate immune system, prior to the phase of extensive growth of the newly generated fibres [5, 6].
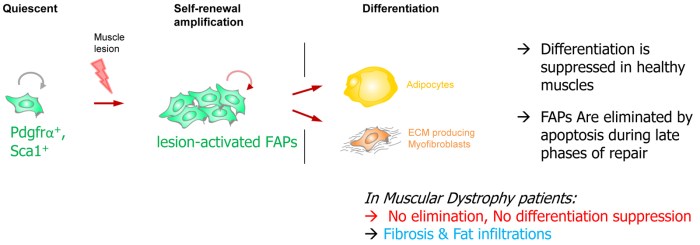
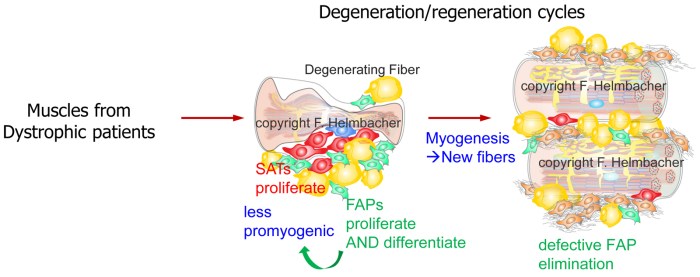
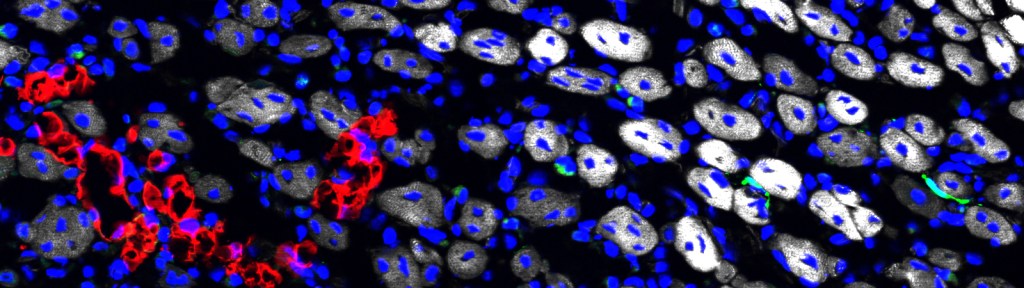
Little is known on the mechanisms suppressing FAP differentiation in time and space. Interestingly, fibrosis and adipose infiltrations (products of FAP differentiation) are symptoms common to several muscular dystrophies [7, 8]. Thus, irrespective of their respective genetic cause, these pathological contexts lead to unleashed FAP differentiation and defective elimination. Preventing fibrosis has become a major goal in the field of muscle pathologies, with emphasis on inhibition of the TGFβ pathway as the leading known fibrosis inducer. To this aim, a key unmet need is to identify naturally occurring mechanisms that block fibro-adipogenic differentiation in healthy muscles, and to understand what is altered in pathological contexts.
References:
- Uezumi, A., et al. Nat Cell Biol, 2010. 12(2): p. 143-52.
- Joe, A.W., et al. Nat Cell Biol, 2010. 12(2): p. 153-63.
- Lukjanenko, L., et al. Cell Stem Cell, 2019.
- Biferali, B., et al. Front Physiol, 2019. 10: p. 1074.
- Lemos, D.R., et al. Nat Med, 2015. 21(7): p. 786-94.
- Heredia, J.E., et al. Cell, 2013. 153(2): p. 376-88.
- Contreras, O., et al. Cell Tissue Res, 2016. 364(3): p. 647-60.
- Mozzetta, C., et al. EMBO Mol Med, 2013. 5(4): p. 626-39.
See also the review I co-wrote with Sigmar Stricker on this. This review focuses on the cross-talks that govern muscle morphogenesis during development but also during adult muscle regeneration, focusing on signals from connective-tissue (including developmental FAPs and adult FAPs).
- Francoise Helmbacher#, Sigmar Stricker#. Tissue cross talks governing limb muscle development and regeneration (Review article). Seminars in Cell and Developmental Biology, (2020), June 7 |PMID: 32517852 | #: co-corresponding | PDF: Helmbacher & Stricker 2020 |
This was followed by a review on FAPs and their contribution to intermuscular adipose tissue physiology and remodelling, co-written with a group of expert in the field under the leadreship of Osvalo Contreras.
- Marcelo Flores-Opazo, Daniel Kopinke, Françoise Helmbacher, Rodrigo Fernández-Verdejo, Mauro Tuñón-Suárez, Gordon S. Lynch, Osvaldo Contreras. Fibro-adipogenic progenitors in physiological adipogenesis and intermuscular adipose tissue remodeling. Molecular Aspects of Medicine (2024), June, 101277 | PDF | PMID : 38788527 |
Recently, we have identified a novel regulator of intramuscular fibro-adipogenic differentiation, representing a key starting point to shed some light on this process. This came from our work on the FAT1 Cadherin, initially identified for its role in muscle morphogenesis, and for its putative contribution as a modifier gene in FSHD. This led us to ask whether FAT1 was involved in muscle regeneration. We used mesenchyme-specific Fat1 mutant mice, and challenged them with skeletal muscle lesions. While this unexpectedly had minor impact on the efficiency of myogenic regeneration, unlike during development, these mice resulted in exacerbated Fibroadipogenic differentiation and adipose infiltrations after skeletal muscle injury (induced with glycerol, which is permissive to adipogenic differentiation).
- Preprint: Pierre-Antoine Ferracci, Françoise Helmbacher. Fat1 deletion enhances Fibro-Adipogenic Differentiation and Adipogenic expansion following injury in skeletal muscle. biorxiv, (2026). April 2026 | DOI | PDF |
We now aim to identify modulators of fibro-adipogenic differentiation necessary to ensure FAP homeostasis. Taking advantage of several murine models of muscular dystrophies with deregulated FAP homeostasis (DFH) leading to fibrosis and adipose infiltrations, we propose a comprehensive program aiming to identify signals deregulated in DHF models accounting for unleashed fibro-adipogenic differentiation, and to distinguish the most promising therapeutic targets