The assembly of a complex neuromuscular circuit involves parallel development of muscles and of the motor and sensory neuronal connections necessary for locomotor functions. In the past, we focused on motor neurons, whereas in recent years, we progressively shifted towards muscles. Below is a brief outline of our work.
Muscle Morphogenesis
Muscle development involves on one hand the definition of the myogenic lineage, which in the trunk, derives from somites, and on the other hand, the interaction of these myogenic cells with the surrounding mesenchyme, thus allowing the positioning of muscle extremities, and the subsequent formation of tendons and joints, at the interface with bones. Multiple molecular players are key to control the stepwise mechanisms occuring during muscle morphogenesis, with regulatory cascades defining the myogenic fate, multiple cell interactions controling expansion of the stem cell pool and production of multinucleated muscle fibers, and 3D control of muscle shape and growth so as to form the many muscles of each of our limbs. Among the many players, we have studied the role of HGF/Met signaling in controling migration of limb myoblasts, and more recently, we identified the Fat1 Cadherin as a novel key player controling shape and growth of subsets of muscles in the face and shoulder. Phenotypic dissection in mice of the tissue-specific modalities of the functions uncovered complementary activities of Fat1 in the myogenic lineage (controling polarized migration and myogenic growth), and in the connective tissues (necessary for selective muscles to expand and adopt their specific shape).
Selected Publications
- Blog post: Helmbacher F. Unraveling tissue interactions coordinating neuromuscular morphogenesis: a journey through serendipity Blog Post, The Node, 20th June, 2018
- Nathalie Caruso, Angela K. Zimmermann, Tarana Nigam, Celine Becker, Karelia Lipson, Françoise Helmbacher. “An intragenic FAT1 regulatory element deleted in muscular dystrophy patients drives muscle and mesenchyme expression during development” Biorxiv (2022), September 17 | doi: 10.1101/2022.09.14.507898 | PDF |
- Helmbacher, F#, Stricker, S#. Tissue cross talks governing limb muscle development and regeneration (Review article). Seminars in Cell and Developmental Biology, (2020), June 7 |PMID: 32517852 | #: co-corresponding | PDF: Helmbacher & Stricker 2020 |
- Helmbacher F. Tissue-specific activities of the Fat1 cadherin cooperate to control neuromuscular morphogenesis. PLOS Biology (2018) 16(5) e2004734 | PMID: 29768404 | doi: 10.1371/journal.pbio.2004734 | previously posted as preprint: https://doi.org/10.1101/207308 |
- Fan Y, Richelme S, Avazeri E, Helmbacher F, Dono R, and Maina F. Tissue-specific gain of RTK signalling uncovers selective cell vulnerability during embryogenesis. PLOS Genetics (2015) September 22 | PMID : 26393505 | DOI: 10.1371/journal.pgen.1005533 |
- Caruso N, Herberth B, Bartoli M, Puppo F, Dumonceaux J, Zimmermann A, Denadai S, Lebossé M, Roche S, Geng L, Magdinier F, Attarian S, Bernard R, Maina F, Levy N, Helmbacher F. Deregulation of the protocadherin gene FAT1 alters muscle shapes: implications for the pathogenesis of facioscapulohumeral dystrophy. PLoS Genet. (2013) Jun;9(6):e1003550. | PMID: 23785297 | doi: 10.1371/journal.pgen.1003550 |
Motor neuron specification
The original focus of the team was essentially related to the aspect of neural diversification among motor neurons, and the developmental mechanisms underlying this diversification, as well as the consequent topographical organization of motor projections towards the muscle targets
Each class of MNs can be recognized not only by its position and the position of its target muscle in the periphery, but also by their molecular characteristics. These are encoded through the expression of selective combinations of transcription factors, each of which controls the expression of a number of downstream effectors, among which axon guidance receptors. Classical studies by experimental biologists had established that Motor neurons become progressively specified to carry the intrinsic knowledge of their anatomical and molecular specificity. Acquisition of such characteristics is specified in nascent motor neurons by environmental signals. One of our long standing interest is to understand the mechanisms governing acquisition of motor neuron cell fates, and in particular in exploring those specified by interaction with peripheral structures.
Selected Publications
- Helmbacher F. Tissue-specific activities of the Fat1 cadherin cooperate to control neuromuscular morphogenesis. PLOS Biology (2018) 16(5) e2004734 | PMID: 29768404 | doi: 10.1371/journal.pbio.2004734 | previously posted as preprint: https://doi.org/10.1101/207308 |
- Caruso N., Herberth B., Lamballe F., Arce-Gorvel V., Maina F., and Helmbacher F. Plasticity versus specificity in RTK signalling modalities for distinct biological outcomes in motor neurons. BMC Biology (2014) 12:56 | PMID:25124859 | doi:10.1186/s12915-014-0056-6 |
- Helmbacher F, Dessaud E, Arber S, Delapeyriere O, Henderson CE, Klein R and Maina F. Met Signaling Is Required for Recruitment of Motor Neurons to PEA3-Positive Motor Pools. Neuron (2003) 39, 767-77. | PMID: 12948444 | doi: 10.1016/S0896-6273(03)00493-8 |
- Blog post: Helmbacher F. Unraveling tissue interactions coordinating neuromuscular morphogenesis: a journey through serendipity Blog Post, The Node, 20th June, 2018
Motor Axon Guidance
Establishment of the topographic innervation of limb muscles by motor neurons involves as series of binary decision between two alternative axonal trajectories, during which growth cones of each motor neuron subtype evaluate peripheral signals and chose their appropriate trajectory to reach their specific muscle target.
This has long been my favorite topic after the initial discovery that the EphA4 tyrosine kinase receptor played an essential role in motor axon pathway selection in the limb (Helmbacher et al., 2000), and that this particular guidance decision represented an excellent model system to study.
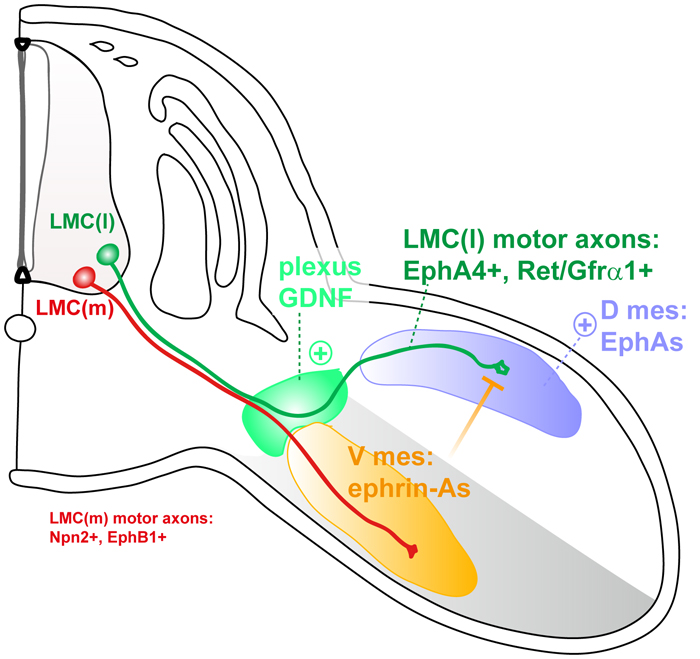
Control of Dorso-ventral pathway selection by limb-innervating Motor axons: Schematic model representing a cross section of a mouse embryo at around E11.5, representing limb-innervating motor neurons in the spinal cord, and the peripheral trajectories of their axons, with some of the molecular players involved. LMCl axons (green) project to the dorsal part of the limb bud. This dorsal trajectory is ensured by the combinatorial activities of several overlapping systems, including EphrinAs/EphA4, GDNF/Ret/Gfra1, and more recently Celsr3/Fzd3. Ventral trajectory of LMC(m) axons is governed by ephrinBs/EphBs and Sema/Npn signaling.
Selected Publications
- Chai G, Zhou L, Manto M, Helmbacher F, Clotman F, Goffinet AM, Tissir F. Celsr3 is required in motor neurons to steer their axons in the hindlimb. Nat. Neuroscience (2014) Aug 10. | PMID: 25108913 | doi:10.1038/nn.3784.
- Charoy C, Nawabi H, Reynaud F, Derrington E, Bozon M, Wright K, Falk J, Helmbacher F, Kindbeiter K, Castellani V. gdnf Activates Midline Repulsion by Semaphorin3B via NCAM during Commissural Axon Guidance. Neuron. (2012) Sep 20;75(6) | PMID: 22998873 |
- Kramer E*, Knott L*, Su F, Dessaud E, Krull CE, Helmbacher F*, and Klein R*. Cooperation between GDNF/Ret and ephrinA/EphA4 signals for motor axon pathway selection in the limb. Neuron (2006). April 6, 50(1):35-47. | PMID: 16600854 | doi: 10.1016/j.neuron.2006.02.020 | (*equal contributions).
- Helmbacher, F, Schneider-Maunoury, S, Topilko, P, Tiret, L and Charnay, P. Targeting of the EphA4 tyrosine kinase receptor affects dorsal/ventral pathfinding of limb motor axons. Development (2000) 127, 3313-24. | PMID : 10887087 |