I’m delighted to share that the manuscript “Astrocyte-intrinsic and extrinsic Fat1 activities regulate astrocyte development and angiogenesis in the retina” is now published in Development (2022) | doi:10.1242/dev.192047 | PDF |
This study uncovers astrocyte-intrinsic and extrinsic Fat1 activities that influence astrocyte migration polarity, proliferation and maturation, the disruption of which impacts retinal vascular development and maintenance of vascular architecture.
Here are a few elements to understand the story and its implications better
Polarized/planar Astrocyte progenitor migration and Retinal Angiogenesis
Endothelial cells (ECs) are the buidling blocks of blood vessels, which assemble and grow through a process called angiogenesis [1]. The mechanisms of angiogenesis are common to many organs. However, some organ-specific adjustments require that ECs cross-talk with organ-specific cellular players. The retina is an excellent example of such interactions between ECs and organ-specific cell types such as astrocytes or neurons [1, 2].
Retinal blood vessels grow from the center to the periphery, along the innermost retinal layer, during the first week after birth in mice. They are preceded by migrating astrocyte progenitor cells, which form a carpet onto which ECs migrate [2]. The two migration events are coupled by known mechanisms involving patterned regulation of angiogenic signals produced by astrocyte progenitors in hypoxic areas, and inhibited by oxygen in blood-perfused areas [2]. Thus, any alteration of astrocyte migration also interferes with angiogenic progression.
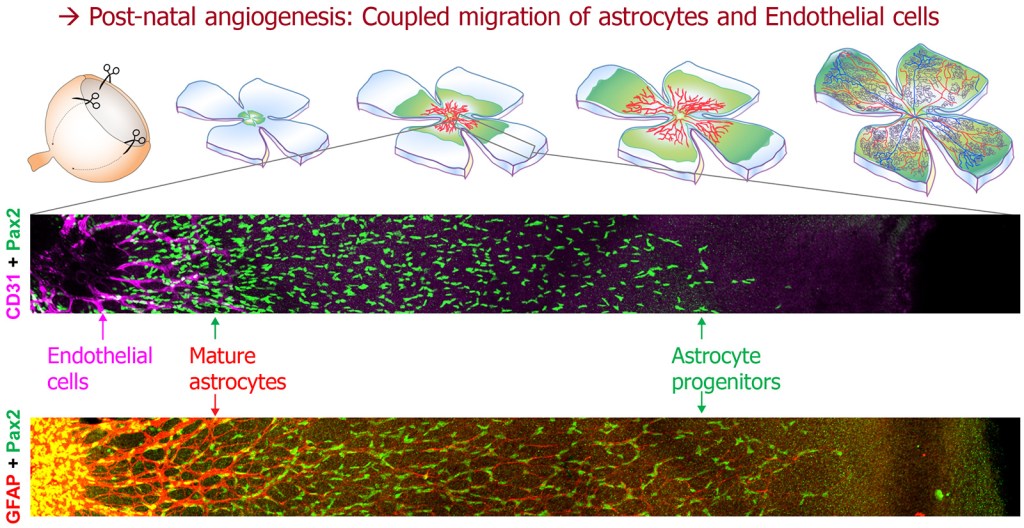
Astrocytes migrate collectively in a polarized manner, headed in a radial/outward direction [3]. This represents a fascinating example of planar polarized migration, raising the possibility that pathways controlling planar cell polarity or polarized migration could interfere with astrocyte progression and likewise with retinal angiogenesis. Our new study shows that one such regulator of tissue morphogenesis, the FAT1 cadherin, influences retinal vascular development by modulating astrocyte migration polarity and maturation [4].
Fat1 modulates retinal astrocyte migration and maturation
In the postnatal retina, Fat1 expression is mainly detected in astrocytes, mueller glia, and subsets of neurons, but not in endothelial cells. Fat1 ablation was first driven with Nestin-cre in the neural lineage, simultaneously targeting astrocytes, Mueller glia, neurons. This interferes with the progression of blood vessel formation in the postnatal retina, and leads to persistent vascular phenotypes in the adult retina [4]. This was associated with a loss of the preferential outward orientation of astrocyte progenitors, and with altered maturation, as seen with GFAP and PDGFRα expression.
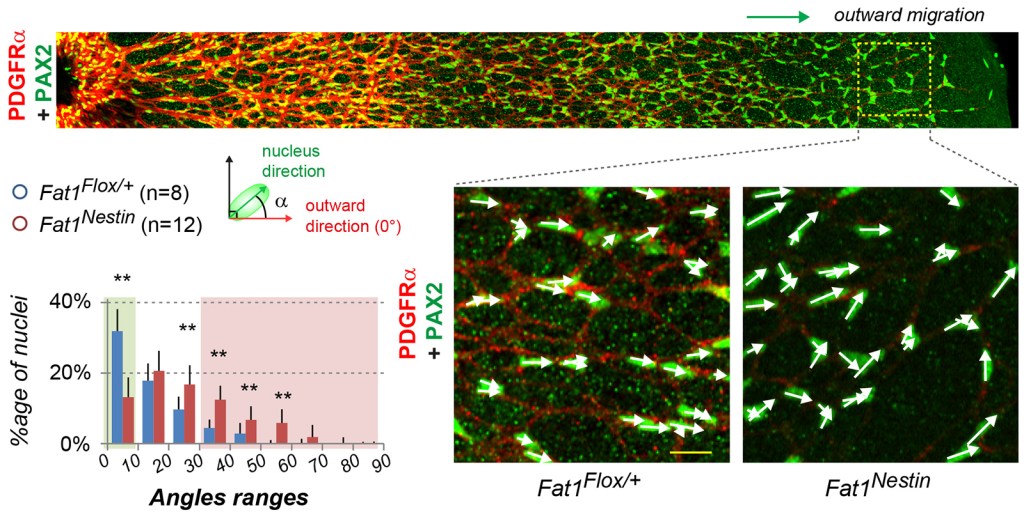
Impaired astrocyte migration and polarity were not associated with alterations of retinal ganglion cell axonal trajectories or of the inner limiting membrane. In contrast, inducible Fat1 ablation in postnatal astrocytes was sufficient to alter their migration polarity and proliferation.
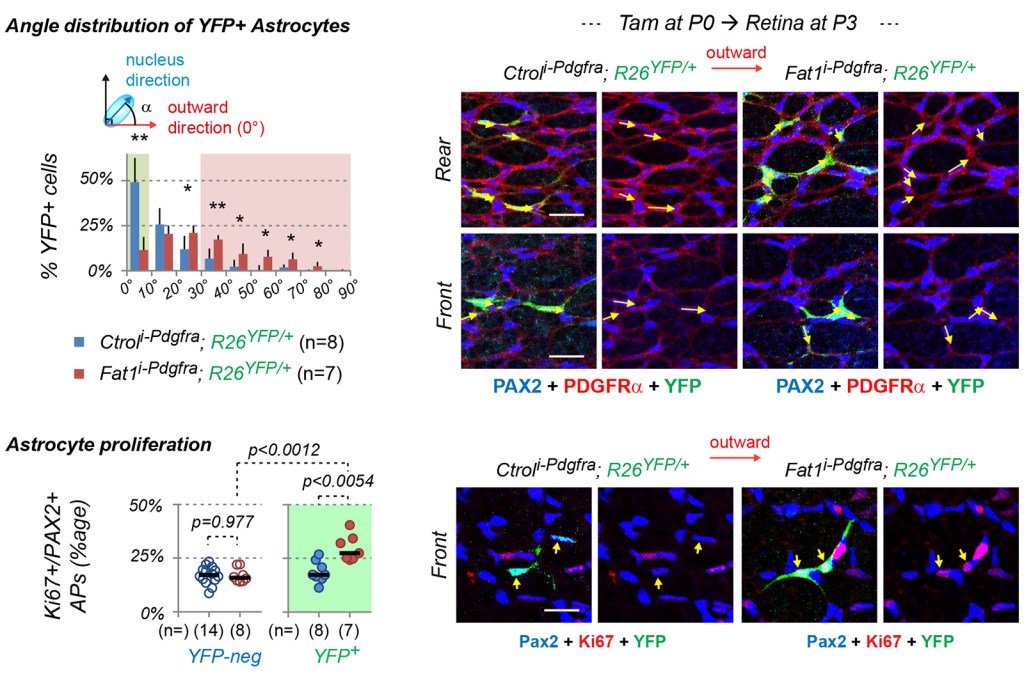
Altogether, this study uncovers astrocyte-intrinsic and extrinsic Fat1 activities that influence astrocyte migration polarity, proliferation and maturation, disruption of which impacts retinal vascular development and maintenance.
Why study retina?
Our initial interest for retinal vasculature stemmed from the potential link between FAT1 and FSHD we identified a few years ago. Aside from muscular symptoms, a number of FSHD patients also present with eye vascular symptoms, collectively referred to as COAT’s symptoms. Although Fat1-deficient mice indeed exhibited retinal vascular phenotypes [5], the underlying biology behind this phenotype was had not been characterized. There is an interesting parallel between the coupled astrocyte/endothelial cell migration events that occur in the postnatal retina and the coupling between myogenic cell migration, myofiber growth and axonal growth that drives neuromuscular development [6].
References
- Paredes, I. Himmels, P. and Ruiz de Almodovar, C. (2018). Neurovascular Communications during CNS Development. Dev Cell 45, 10-32.
- Tao, C. and Zhang, X. (2014). Development of astrocytes in the vertebrate eye. Dev Dyn 243, 1501-1510.
- O’Sullivan, M. L., Punal, V. M., Kerstein, P. C., Brzezinski, J. A. t., Glaser, T., Wright, K. M. and Kay, J. N. (2017). Astrocytes follow ganglion cell axons to establish an angiogenic template during retinal development. Glia 65, 1697-1716
- Helmbacher, F. (2022) Astrocyte-intrinsic and extrinsic Fat1 activities regulate astrocyte development and angiogenesis in the retina. Development 15 January 2022; 149 (2): dev192047 | PDF | PMID: 35050341 |
- Caruso N., Herberth B., Bartoli M., Puppo F., Dumonceaux J., Zimmermann A., Denadai S., Lebossé M., Roche S., Geng L., Magdinier F., Attarian S., Bernard R., Maina F., Levy N. and Helmbacher F. (2013). Deregulation of the protocadherin gene FAT1 alters muscle shapes: implications for the pathogenesis of Facioscapulohumeral dystrophy. PLoS Genet.
- Helmbacher, F. (2018). Tissue-specific activities of the Fat1 cadherin cooperate to control neuromuscular morphogenesis. PLoS Biol 16, e2004734